- To increase the number of conduction-band electrons in intrinsic silicon, pentavalent impurity atoms are added;
- These are atoms with five valence electrons such as arsenic (As), phosphorus (P), bismuth (Bi), and antimony (Sb);
- As illustrated in Figure 1–17, each pentavalent atom (antimony, in this case) forms covalent bonds with four adjacent silicon atoms;
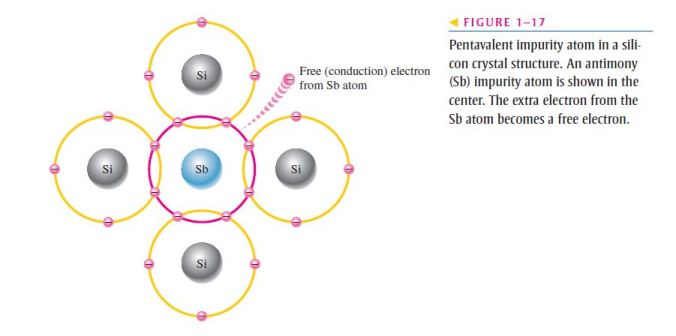
- Four of the antimony atom’s valence electrons are used to form the covalent bonds with silicon atoms, leaving one extra electron;
- This extra electron becomes a conduction electron because it is not involved in bonding;
- Because the pentavalent atom gives up an electron, it is often called a donor atom;
- The number of conduction electrons can be carefully controlled by the number of impurity atoms added to the silicon;
- A conduction electron created by this doping process does not leave a hole in the valence band because it is in excess of the number required to fill the valence band;
Majority and Minority Carriers
- Since most of the current carriers are electrons, silicon (or germanium) doped with pentavalent atoms is an n-type semiconductor (the n stands for the negative charge on an electron);
- The electrons are called the majority carriers in n-type material;
- Although the majority of current carriers in n-type material are electrons, there are also a few holes that are created when electron-hole pairs are thermally generated;
- These holes are not produced by the addition of the pentavalent impurity atoms;
- Holes in an n-type material are called minority carriers;