- Recall that the valence shell of an atom represents a band of energy levels and that the valence electrons are confined to that band;
- When an electron acquires enough additional energy, it can leave the valence shell, become a free electron, and exist in what is known as the conduction band;
- The difference in energy between the valence band and the conduction band is called an energy gap or band gap;
- This is the amount of energy that a valence electron must have in order to jump from the valence band to the conduction band;
- Once in the conduction band, the electron is free to move throughout the material and is not tied to any given atom;
- Figure 1–7 shows energy diagrams for insulators, semiconductors, and conductors;
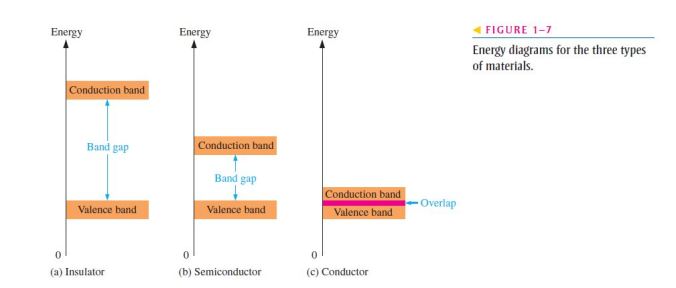
- The energy gap or band gap is the difference between two energy levels and is “not allowed” in quantum theory;
- It is a region in insulators and semiconductors where no electron states exist;
- Although an electron may not exist in this region, it can “jump” across it under certain conditions;
- For insulators, the gap can be crossed only when breakdown conditions occur—as when a very high voltage is applied across the material;
- The band gap is illustrated in Figure 1–7(a) for insulators;
- In semiconductors the band gap is smaller, allowing an electron in the valence band to jump into the conduction band if it absorbs a photon;
- The band gap depends on the semiconductor material. This is illustrated in Figure 1-7(b);
- In conductors, the conduction band and valence band overlap, so there is no gap, as shown in Figure 1–7(c);
- This means that electrons in the valence band move freely into the conduction band, so there are always electrons available as free electrons;